 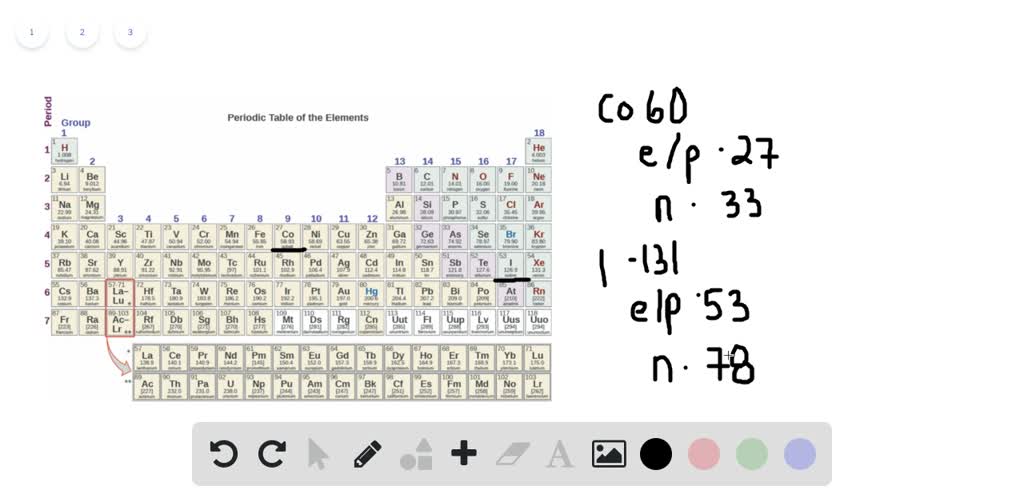
In scrap metal is responsible for the radioactivity found in several iron-based products. Steel contamination Ĭobalt is an element used to make steel. Over time, the absorbed cobalt is eliminated in urine. The remainder is taken up by tissues, mainly the liver, kidneys, and bones, where the prolonged exposure to gamma radiation can cause cancer. 59Īfter entering a living mammal (such as a human being), some of the 60 Still, over 40% of all single-use medical devices are sterilized using 60Ĭo from Bruce nuclear generating station. The cobalt targets are substituted here for a small number of fuel assemblies. In the United States, it is now being produced in a BWR at Hope Creek Nuclear Generating Station. , by substituting the control rods with cobalt rods. The CANDU reactors can be used to activate 59 Californium-252, moderated through water, can be used for this purpose, as can the neutron flux in a nuclear reactor. Source for sterilizing screwflies in the 1959 Screwworm Eradication Program. No country is known to have done any serious development of this type of weapon. When the bomb exploded, the excess neutrons from the nuclear fission would irradiate the cobalt and transmute it into 60 In one hypothetical design, the tamper of the weapon would be made of 59 Nuclear fallout, rendering them uninhabitable. Ĭobalt has been discussed as a " salting" element to add to nuclear weapons, to produce a cobalt bomb, an extremely "dirty" weapon which would contaminate large areas with 60 As a radiation source for food irradiation and blood irradiation.Radiation source for pest insect sterilization.Radiation source for leveling devices and thickness gauges.Radiation source for industrial radiography.Cobalt therapy, using beams of gamma rays from 60 Radiation source for medical radiotherapy.As a tracer for cobalt in chemical reactions.The physical properties of cobalt such as resistance to bulk oxidation and low solubility in water give some advantages in safety in the case of a containment breach over some other gamma sources such as caesium-137. The β-decay energy is low and easily shielded however, the gamma-ray emission lines have energies around 1.3 MeV, and are highly penetrating. Is that it is a high-intensity gamma-ray emitter with a relatively long half-life, 5.27 years, compared to other gamma ray sources of similar intensity. Prototype irradiator for food irradiation to prevent spoilage, 1984. , emitting 58.6 keV gamma rays, or with a low probability (0.22%) by β-decay into 60 Internal conversion energies are well below the main energy levels. In the diagram the two important ones are marked. Energy transfers between the three levels generate six different gamma-ray frequencies. The probability for population of the middle energy level of 2.1 MeV by β-decay is 0.0022%, with a maximum energy of 665.26 keV. The diagram shows a (simplified) decay scheme of 60 This amounts to nearly 20 watts per gram, nearly 30 times larger than that of 238 The high γ-energies result in a significant mass difference between 60 Devices for nondestructive material testing use sources with activities of 1 TBq and more. Test sources, such as those used for school experiments, have an activity of <100 kBq. Reduces the distance to a few millimeters, and the same dose is achieved within seconds. 
, generates a dose of 1 mSv at one meter distance within one hour. Source with an activity of 2.8 GBq, which is equivalent to 60 μg of pure 60 This allows calculation of the equivalent dose, which depends on distance and activity. It is equal to 0.35 mSv/(GBq h) at one meter from the source. The absorbed dose constant is related to the decay energy and time. The activated nickel nucleus emits two gamma rays with energies of 1.17 and 1.33 MeV, hence the overall equation of the nuclear reaction (activation and decay) is:Ĭorresponding to its half-life, the radioactive activity of one gram of 60 Undergoes beta decay to the stable isotope nickel-60 ( 60 The simplest case of the latter would result from the activation of 58 Is largely the result of multiple stages of neutron activation of iron isotopes in the reactor's steel structures via the creation of its 59 In the latter case (in the absence of added cobalt) the incidentally produced 60 Measurable quantities are also produced as a by-product of typical nuclear power plant operation and may be detected externally when leaks occur. Deliberate industrial production depends on neutron activation of bulk samples of the monoisotopic and mononuclidic cobalt isotope 59 : 39 It is produced artificially in nuclear reactors. Cobalt-60, 60CoĬobalt-60 ( 60Co) is a synthetic radioactive isotope of cobalt with a half-life of 5.2713 years. For the state highway in Colorado, see Colorado State Highway 60. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
